Upneeq
*In order to purchase this product you must also schedule a Virtual Consultation.
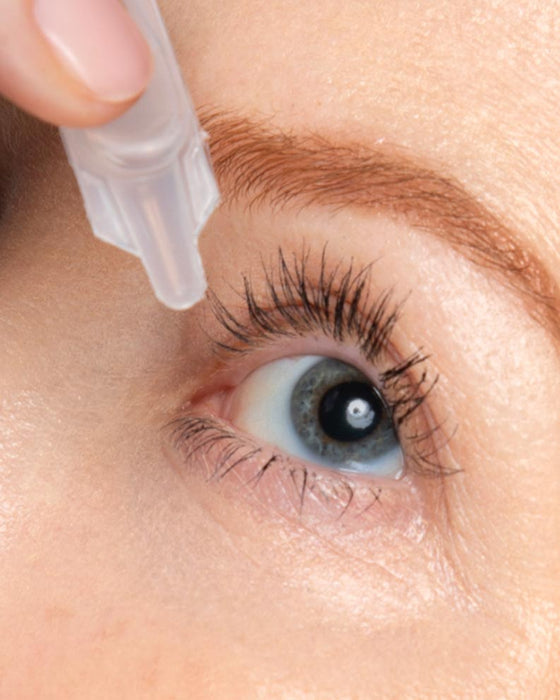
Imagine an EYE-OPENING Lift with a Daily Drop of Upneeq®
The only FDA-approved prescription eyedrop for acquired ptosis (low-lying lids) that lifts your upper eyelids to open your eyes.
This is the perfect combination product for any eye treatment (eye-lid surgery, laser treatments, thread lifts, fillers, plasma pen, chemical peels, AccuTite, Ultherapy, Renuvion, etc.).
This package contains 30 single-use containers in individual, child-resistant foil pouches .